 
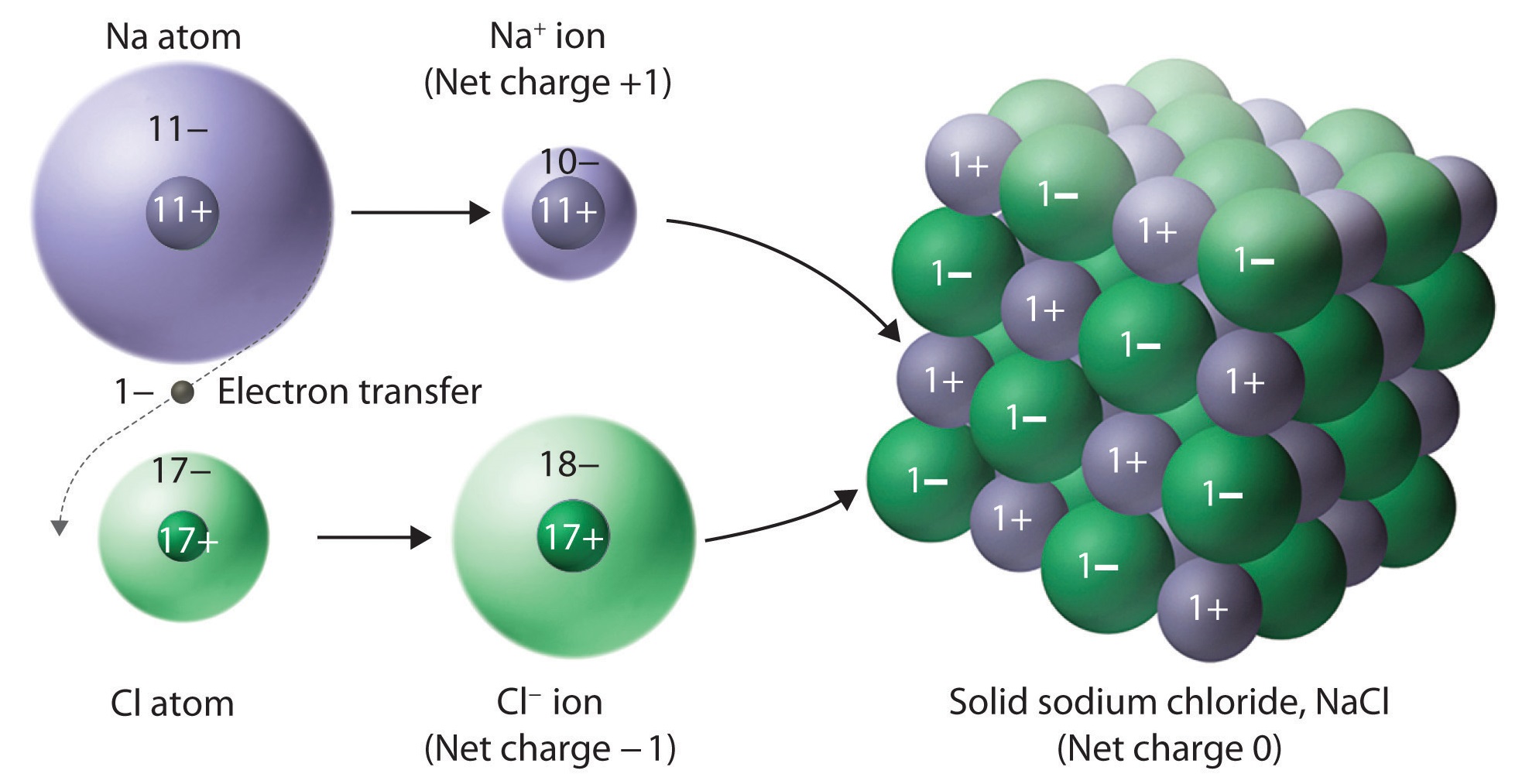
Compounds that do not contain ions, but instead consist of atoms bonded tightly together in molecules (uncharged groups of atoms that behave as a single unit), are called covalent compounds. Ionic compounds generally form from metals and nonmetals. Ions can be either monatomic (containing only one atom) or polyatomic (containing more than one atom).Ĭompounds that contain ions are called ionic compounds. Positively charged ions are called cations, and negatively charged ions are called anions. Thus, nonmetals tend to form negative ions. Similarly, nonmetals (especially those in groups 16 and 17, and, to a lesser extent, those in Group 15) can gain the number of electrons needed to provide atoms with the same number of electrons as in the next noble gas in the periodic table. By this means, a positively charged ion is formed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
